Streamlined and Intelligent Documentation
The newly developed COMOS-based GMP Document Management solution enables automatic creation, validation, and control of plant documentation. Users can instantly populate documents from preformatted Excel templates, supported by an auto-nomenclature system aligned with plant hierarchy. This approach reduces manual errors and accelerates document preparation during engineering and qualification phases.
Compliant and Secure Document Lifecycle
To meet GMP and 21 CFR Part 11 compliance, the system features validation and approval cycles, digital signatures, and intelligent watermarks that dynamically indicate the document’s status and version. The bottom banner automatically updates when a user prints any document, ensuring traceability and compliance in every stage of document handling.
Seamless Integration and Traceability
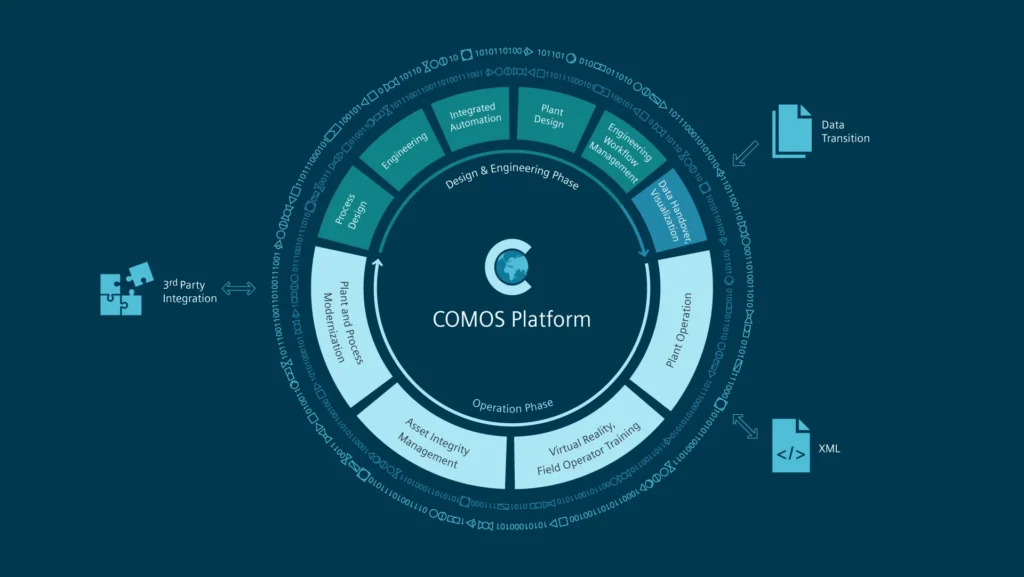
Pre-defined roles and integration with Active Directory make user access management effortless. IQ/OQ/PQ cycles can be triggered through user-defined events, while auto-generated reports simplify review, indexing, and audit trail management. The unified COMOS database hosts intelligent drawings — including BFDs, PFDs, P&IDs, datasheets, layouts, SLDs, and wiring diagrams — all linked to an integrated 3D visualization environment.
Key Highlights
- Plant hierarchy with automated nomenclature
- Excel import plugin for document population
- Validation and approval workflows
- Active Directory–based role management
- Intelligent watermarks and print banners
- Digital signature placeholders
- IQ/OQ/PQ cycle automation with user-defined triggers
- Automated review, index, and audit reports
- Single database for intelligent drawings
- Built-in 3D visualization
Learn more about our COMOS-based GMP Document Management Solution → GMP document Management – NM Technocrafts