Pharma API & Formulation Manufacturer
- Home
- Case Studies
- GMP document Management
GMP document Management
This is a global pharmaceutical company based in India, known for its role in the development, manufacturing, and marketing of a wide range of pharmaceutical products. The company has a diverse portfolio that includes generic and branded medications. It is known for its strong research and development capabilities, allowing it to innovate and expand its product offerings. They reported an annual turnover or revenue of approximately INR 25,500 crore (or around USD 3.5 billion)
Problem Statement
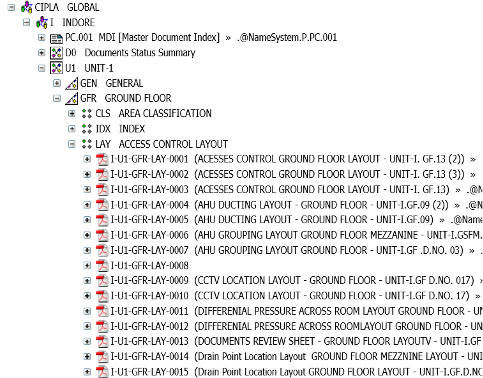
GMP Document Management
- This client was looking for a centralized Document Management System to maintain GMP documents for their manufacturing facilities (close to 52 Units with Approx. ~ 12K Drawings)
- Solution to have following capabilities:
- Auto-Drawing number generation as per Site à Location à Building à Floor à Room
- Approval workflows including final signing off by QA team
- Digital Signature with validation
- Auto-generation of Review sheet for every document
- Auto-generation of Drawing List with Audit train Report
- Only Latest document to be visible to user
- Watermark on drawings for Approved and Uncontrolled copy
- User Name, Location, date and time to be displayed on real time basis on drawings printed by Users
Our Solution
COMOS as centralized solution for maintaining all plant documents with proper Hierarchy as shown below: